Cobalt Oxide Co3O4 powder

Specification
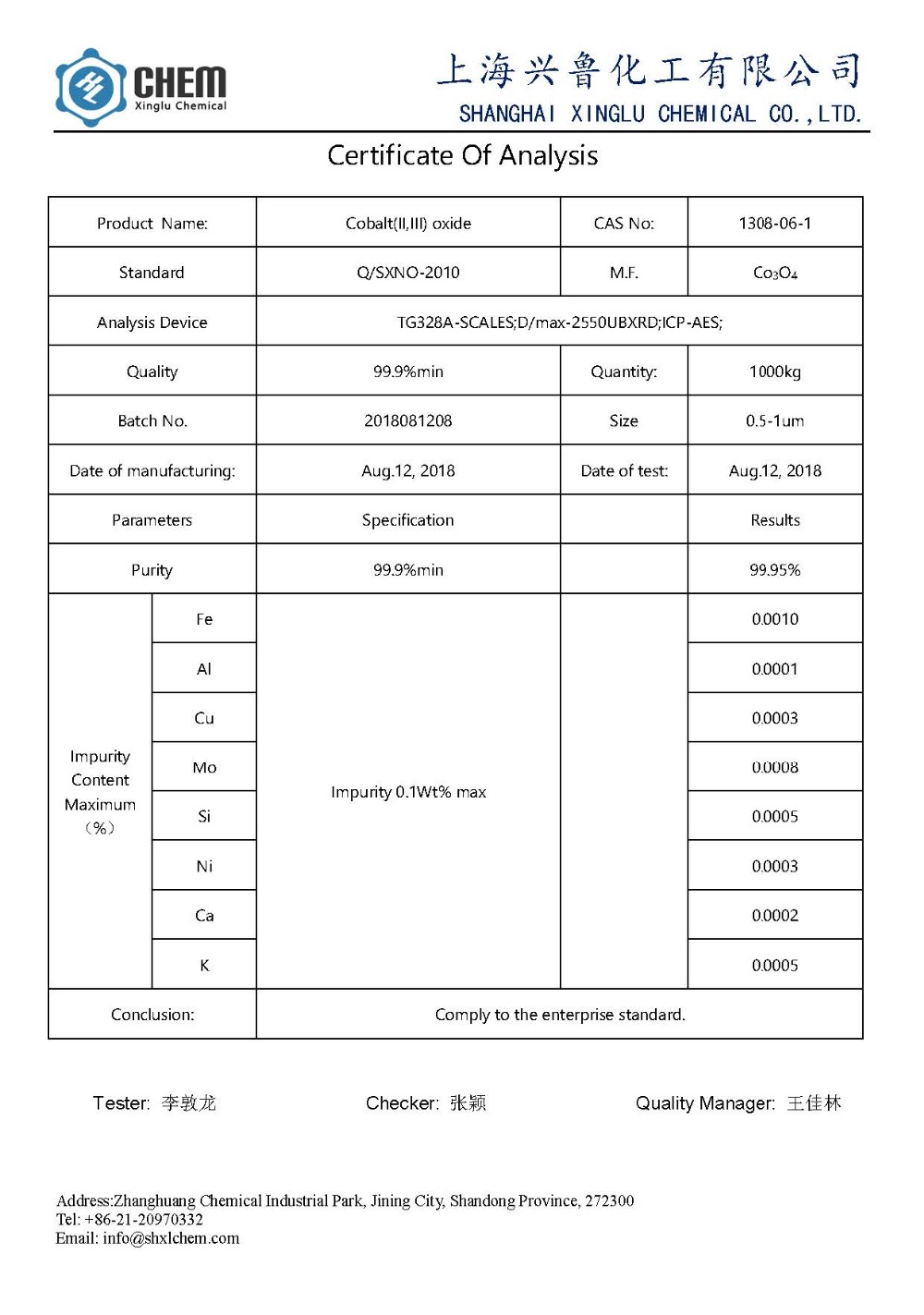
1.Name: nano Cobalt Oxide Co3O4 powder
2.Purity: 99.9% min
3.Appearacne: gray black powder
4.Particle size: 50nm
5.SSA: 30-80 m2/g
Properties:
Exposure to air, easy to absorb moisture, but does not generate water compounds. It is soluble in nitric acid. When heated to above 1200 oC, nano-cobalt oxide will be broken down into sub-cobalt oxide. In the hydrogen flame, nano-cobalt oxide is heated to 900 oC, it will be transformed into metal cobalt. Cobalt(II,III) oxide is chemical compound with the formula Co3O4. It is a black solid, and a mixed valence compound, containing both Co(II) and Co(III) oxidation states. It can be formulated as CoIICoIII2O4 or CoO.Co2O3. Cobalt(II) oxide, CoO, converts to Co3O4 if heated to around 600-700 °C in air. Above 900 °C, CoO is stable.
Application:
Catalysis, superconductors, ceramics and other fields as an important inorganic materials; As catalyst and catalyst carrier and the electrode active material; For glass, porcelain colorants and pigments; Chemical industry oxidant and a catalyst for organic synthesis; Senior goggles and other filter materials; Carbide; Temperature and gas sensors; For the semiconductor industry, electronic ceramics, lithium ion battery electrode materials, magnetic materials; Electrochromic devices; Enamels; Grinding wheels; Heterogeneous catalysts; Solar energy absorbers....
Certificate:

What we can provide: